Characterization of a novel lyophilized chitosan hydrogel complex for the controlled release of a highly water soluble drug, niacinamide
Keywords:
Chitosan hydrogel complex, controlled drug delivery, niacinamide, lyophilization, Carbopol 974P-NF, HPMC K4MAbstract
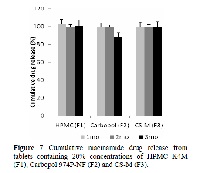
The purpose of this research was to prepare and characterize a novel lyophilized chitosan-based hydrogel complex (termed CS-M) for controlled drug delivery applications using a highly water soluble model drug, niacinamide. Characterization studies were undertaken to evaluate the physical-chemical properties, polymer swelling, in vitro controlled release kinetics, tissue bioadhesion, intestinal permeability and stability of the novel chitosan complex. Additionally, a comparative analysis was conducted with commercial polymers namely, Carbopol 974P-NF® and hydroxypropyl methylcellulose (HPMC K4M). FT–IR and 1H NMR studies confirmed that despite alteration in physical structure and morphology of the chitosan complex the chemical properties remained unchanged, when compared to the parent chitosan compound. Polymer swelling studies showed consistency in the structural integrity and water uptake of CS-M compared to other polymers which showed inconsistent swelling and disintegration behavior over a 5 h period. In vitro controlled release profiles of CS-M showed a slower, more controlled release rate of niacinamide than other polymers indicating the influence of polymer swelling capacity on water uptake and subsequent drug release. CS-M demonstrated an overall increase in bioadhesion to intestinal tissue when compared to commercial polymers at same concentrations. Similarly, drug transport through everted sac intestinal tissue showed enhanced absorption properties of CS-M when compared to other polymers. Finally, a 3 month accelerated stability study showed all polymers including CS-M to be stable when formulated with niacinamide. Overall, the modified chitosan-based hydrogel polymer, CS-M, demonstrated enhanced characteristics indicating its potential to be used as a controlled release excipient in oral drug formulations.
References
Suh JKF, Matthew HWT. Application of
chitosan-based polysaccharide biomaterials in
cartilage tissue engineering: A review. Biomat,
; 21: 2589-2598.
Khoe E, Lim LY. Implantable applications of
chitin and chitosan. Biomat, 2003; 24: 2339-2349.
Anthonsen MW, Varum KM, Smidsrod O.
Solution properties of chitosans: Conformation
and chain stiffness of chitosans with different
degree of n-acetylation, Carbohydr Poly, 1993;
: 193-201.
Dodane V, Khan AM, Merwin JR. Effect of
chitosan epithelial permeability and structure. Int
J Pharm, 1999; 182: 21-32.
Illum L. Chitosan and its use as a pharmaceutical
excipient. Pharm Res, 1998; 15: 1326-1331.
Bernkop-Schnurch A, Hornof M, Guggi D.
Thiolated Chitosans. Eur J Pharm Biopharm,
; 57: 9-17.
du Plessis LH, Kotze AF, Junginger HE. Nasal
and rectal delivery of insulin with chitosan and Ntrimethyl chitosan chloride. Drug Del, 2010;
(6): 399-407.
Prabhu S, Ma C, Ortega M. Novel combinations
of rate controlling polymers for the release of
leuprolide acetate in the colon. Drug Del, 2008;
: 119-125.
Prada P, de la Torre P, Torrado S. Chitosanpoly(acrylic) acid polyionic complex: In vivo
study to demonstrate prolonged gastric retention.
Biomat, 2004; 25: 917-923.
Quintanar-Guerrero D, Villalobos-Garcia R,
Alvarez-Colin E, Cornejo-Bravo JM. In vitro
evaluation of the bioadhesive properties of
hydrophobic polybasic gels containing N,Ndimethylaminoethyl methacrylate co-methyl
methacrylate. Biomat, 2001; 22: 957-961.
Mariappan TT, Singh S. Evidence if efflux
mediated and saturable absorption of rifampicin
in rat intestine using the ligated loop and everted
gut sac technique. Macromol Pharm, 2004; 5:
-367.
Ponchel G, Touchard F, Duchene D, Pappas NA.
Bioadhesive analysis of controlled release
systems I. Fracture and interpenetration analysis
in poly(acrylic acid) containing systems. J Contr
Rel, 1987; 5: 129-141.
Sannan T, Kurita K, Iwakura Y. Studies on chitin,
Effect of deacetylation on solubility. Macromol
Chem, 1976; 177: 3589-3600.
Dai Y-N, Li P, Zhang JP, Wang AQ, Wei Q.
Swelling characteristics and drug delivery
properties of nifedipine-loaded pH sensitive
alginate-chitosan hydrogel beads. J Biomed Mater
Res B, 2008; 86: 493-500.
Siepmann J, Peppas NA. Modeling of drug
release from delivery systems based on
hydroxypropyl methylcellulose (HPMC K4M).
Adv Drug Del Rev, 2001; 48: 139-157.
Lee PI, Peppas NA. Prediction of polymer
dissolution in swellable controlled-release
systems. J Contr Rel, 1987; 6: 207-215.
Gonzalez-Rodriguez ML, Maestrelli F, Rabasco
AM. In vitro release of sodium diclofenac from a
central core matrix tablet aimed for colonic drug
delivery. Eur J Pharm Sci, 2003; 20: 125-131.
Amiji MM. Pyrene fluorescence study of chitosan
self-association in aqueous solution, Carbohydr
Poly, 1995; 26: 211-213.
Diez-Sales O. In vitro percutaneous penetration of
acyclovir from solvent systems and Carbopol
-P hydrogels: Influence of propylene glycol. J
Pharm Sci, 2005; 94: 1039-1047.
Sung KC, Nixon PR, Skoug JW, Ju R, Gao P,
Topp EM, Patel MV. Effect of formulation
variables on drug and polymer release from
HPMC K4M-based matrix tablets. Int J Pharm,
; 146: 53-60.
Lehr CM, Bouwstra JA, Schacht EH, Junginger
HE. In vitro evaluation of mucoadhesive
properties of chitosan and some other natural
polymers. Int J Pharm, 1992; 78: 43-48.
Artursson P, Lindmark T, Davis SS, Ilum L.
Effect of chitosan on the permeability of
monolayers of intestinal epithelial cells (caco-2).
Pharm Res, 1994; 11: 1358-1361.
Schipper NGM, Varum KM, Artursson P.
Chitosan as absorption enhancers for poorly
absorbable drugs I: Influence if molecular weight
and degree of acetylation on drug transport
across the human intestinal epithelial (caco-2)
cells. Pharm Res, 1996; 13: 1686-1692. 9; 44:
-59.