Taste masking of Lornoxicam by polymer carrier system and formulation of oral disintegrating tablets
Keywords:
Oral disintegrating tablet, Lornoxicam, Eudragit EPO, Super disintegrating agents, Taste maskingAbstract
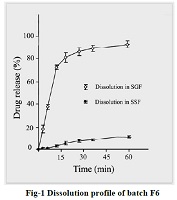
Lornoxicam is a non steroidal anti-inflammatory drug with analgesic properties and belongs to the class oxicams. It is extremely bitter in taste. The purpose of this research was to develop a bitterless oral disintegrating tablet of Lornoxicam. Taste masking was done by complexing Lornoxicam with aminoalkyl methacrylate copolymer (Eudragit EPO) in different ratios. In vitro release profile obtained at pH 6.2 indicate that perceivable amount of drug will not be released in saliva while high percentage release (more than 80 % in 30 mins.) would be obtained at acidic pH 1.2 of the stomach. Three super disintegrants were used while preparing the tablets e.g. sodium starch glycolate, crospovidone and crosscarmellose sodium. The tablets were evaluated for different properties like drug content, hardness, friability and disintegration time. The tablets shown good taste and disintegration in oral cavity.
References
Chang R, Guo X, Burnside B, Couch R. A review of fast dissolving tablets. Pharm Tech. (North America). 2000;52-58.
Bi Y, Sunada H, Yonezawa Y, Dayo K, Otsuka A, Iida K. Preparation and evaluation of a compressed tablet rapidly disintegrating in oral cavity. Chem Pharm Bull (Tokyo). 1996;44:2121-2127.
Watanabe Y, Ishikawa Y, Utoguchi N. Matsumoto M. Preparation and evaluation of tablets rapidly disintegrating in saliva containing bitter taste masked granules by the compression method. Chem. Pharm. Bull. 1999; 47:1451-1457.
Kaushik D, Dureja H, Saini TR. Mouth dissolving tablets: A review. Indian Drugs-Bombay. 2004; 41: 187-193.
Balfour JA, Fitton A, Barradell LB. Lornoxicam. A review of its pharmacology and therapeutic potential in the management of painful and inflammatory conditions. Drugs. 1996;51(4):639-57.
Lorenz IH, Egger K, Schubert H, Schnürer C, Tiefenthaler W, Hohlrieder M, Schocke MF, Kremser C, Esterhammer R, Ischebeck A, Moser PL, Kolbitsch C. Lornoxicam characteristically modulates cerebral pain-processing in human volunteers: a functional magnetic resonance imaging study. British Journal of Anaesthesia 2008; 100(6):827-833.
Makooi-Morehead WT, Buehler JD, Landmann, BR. US Patent No., US6238695, 2001.
Watanabe Y., Ishikawa Y., Utoguchi N. and Matsumoto M. Preparation and evaluation of tablets rapidly disintegrating in saliva containing bitter taste masked granules by the compression method. Chem. Pharm. Bull., 1999, 47, 1451-1457.
Cousin G, Bruna E, Gendrot E, US Patent No., US5464632, 1995.
Kibbe A., In; Handbook of Pharmaceutical Excipients, 3rd Edn., The Pharmaceutical Press, London; 2000, p 501.
Goodhart FW, Draper JR, Dancz D, Ninger FC. Evaluation of tablet breaking strength testers. J Pharm Sci 1973; 62(2): 297-304
Sreenivas SA, Gadad AP. Formulation and evaluation of Ondancetron Hcl directly compressed mouth disintegrating tablets. Indian Drugs, 2006; 43(1):35-38.
Banker GS, Anderson NR. In; Lachman, L, Lieberman, HA, Kanig, JL, Eds., The Theory and Practice of Industrial Pharmacy, 3rd Edn., Lea and Febiger, Philadelphia; 1991, p 293.
Aley AM, Semreen M, Qato MK. To produce rapidly disintegrating Tenoxicam tablet via Camphor sublimation. Pharmaceutical Technology, 2005; p 68-78.
Bi Y, Sunanda H, Yonezawa Y, Danjo K, Otsuka A, Iida K. Preparation and evaluation of a compressed tablet rapidly disintegrating in the oral cavity. Chem. Pharm. Bull., 1996, 44: 2121-2127
Reddy LH, Gosh BR. Fast dissolving drug delivery system: A Review of the literature. Indian J Pharm Sci. 2002;64: 331-336.