Demonstration of Multivariate Data Analysis for the Development of nanoemulsions containing Active Herbal Principle of Boswellia serratta for Topical Application
Keywords:
Boswellia serrata extract, nanoemulsion, Pseudo ternary phase diagram, Nanoemulsion based hydrogel, Simplex lattice design, Partial least square analysisAbstract
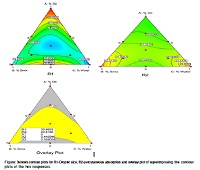
The chemometric techniques have been used to demonstrate the role of nanoemulsion components on release of active herbal principle present in the spray dried Boswellia serratta extract. Isopropyl myristate, Tween 80 and Transcutol PR were selectedafter determining the solubility of boswellic acids (BAs) and used to draw phase diagrams. Simplex lattice mixture design was applied to optimize the percentual composition of nanoemulsions.The Permeability coefficient and droplet size were modelled with set of variables by partial least squares (PLS). Partial least square regression analysis was done through Excel STAT to influence mixture composition on permeation behavior of drug from the nanoemulsion as calculated models revealed good predictive abilities.Ex vivo skin permeation and in vivo anti-inflammatory study were conducted to evaluate the potential of optimized nanoformulations. About 3.25fold increase in flux was seen in case of nanoemulsion, nanogel showed 1.45 fold increase in flux as compared to carbopol gel with highest enhancement ratio 4.57 and 1.59 respectively. Physicochemically stable and non-irritant hydrogel showed significant percentage inhibition in rat paw edema. Our study illustrated scientific and statistical evidencefor the potential of developed nanoemulsion as possible alternative to traditional topical formulations.
References
Gerbeth K, Meins J, Kirste S, Momm F, Schubert-Zsilavecz M, Abdel-Tawab M., Determination of major boswellic acids in plasma by high-pressure liquid chromatography/mass spectrometry. J Pharm Biomed Anal, 56 (2011) 5 998–1005. DOI:10.1016/J.FITOTE.2012.10.009
Adnan Azeem, Mohammad Rizwan, Roop K. Khar, and Sushama Talegaonkar, Nanoemulsion Components Screening and Selection: a Technical Note AAPS PharmSciTech, 10 (2009) 69-76. DOI:10.1208/S12249-008-9178-X
Ajazuddin, s. saraf, Applications of novel drug delivery system for herbal formulations Fitoterapia, 81 (2010) 680-689.
Chaturvedi Mayank, Manish Kumar, Amit Sinhal, Alimuddin Saif, Recent development in novel drug delivery systems of herbal drugs Int J. Green Pharm. 5 (2011) 87-94. DOI: 10.4103/0973-8258.85155
Gould, P.L., Optimization methods for the development of dosage forms. Int. J. Pharm. Tech. Prod. Mfr., 5 (1984) 19-24.
Pattarino F, Marengo E. Gasco M.R. Carpignano R., Experimental design and partial least squares in the study of complex mixtures: microemulsions as drug carriers. Int J Pharm. 91 (1993) 157-165
Sonneville-Aubrun, O., Simonnet, J.-T., L’Alloret, F., Nanoemulsions: a new vehicle for skincare products. Adv. Colloid Interface Sci. 108–109 (2004) 145–149
Chen H, Chang X, Du D, Li J, Xu H, Yang X., Microemulsion-based hydrogel formulation of ibuprofen for topical delivery. Int J Pharm, 52 (2006) 315.
Peltola S, Saarinen‑Savolainen P, Kiesvaara J, Suhonen TM, Urtti A., Microemulsions for topical delivery of estradiol. Int J Pharm, 254 (2003) 99‑107.
Saji Uthaman, Snima, Annapoorna, Ravindranath, & Shanti, Novel Boswellic acids Nanoparticles induces cell death in Prostate cancer cells. J Natural Products, 5 (2012) 100-108.
Goel, Jalees, Mohan, 3-Acetyl-11-keto-b-boswellic acid loaded-polymeric nanomicelles for topical anti-inflammatory and anti-arthritic activity J. Pharm. Pharmacol. 62 (2010) 273–278 DOI : 10.1211/jpp/62.02.0016
Dreher F, Walde P, Walter P, Wehrli E., Interaction of a lecithin microemulsion gel with human stratum corneum and its effect on transdermal transport. J Control Rel. 45 (1997) 131-140.
Eriksson, L., Johansson, E., Wikstrom, C., Mixture design: design generation, PLS analysis, and model usage. Chemomet. Intell. Lab. Syst. 43 (1998) 1–24.
Weiwei Zhu, Aihua Yu, Weihong Wang, Ruiqian Dong, Formulation design of microemulsion for dermal delivery of penciclovir. Int J Pharm, 360 (2008) 184-190.
Wold, H., Soft modeling: The basic design and some extensions. In Joereskog, K.G. and Wold, H. (Eds), Systems under Indirect Observation: Causility-Structure-Prediction. II. North-Holland, Amsterdam, (1982) 1-54.
Geladi, P. and Kowalski, B.R., Partial least-squares regression: a tutorial. Anal, Chim. Aeta 185 (1986) 1-17.
Manne, R., Analysis of two partial least-squares algorithms for multivariate calibration, Chemometrics lntell. Lab. Systems, 2 (1987) 187-197.
Gasco M.R., Pattarino F., Experimental design and partial least squares in Journal of Pharmaceutics, 91 (1993) 157-165
Marengo, E. and Todeschini, R., A fast method for the calculation of partial least-squares coefficients. Chemometrics Intel. Lab. Systems, 12 (1991) 117-120.
Faiyaz Shakeel, Sanjula Baboota, Javed Ali, Nanoemulsions as Vehicles for Transdermal Delivery of Aceclofenac, AAPS PharmSciTech, 8 (2007) E1-E9. DOI:10.1208/PT0804104
Giraldi, T., Perissin, L., Zorzet, S., Rapozzi, V., Ann. N.Y., Stress, melatonin and tumour progression in mice. Acad. Sci. 719 (1994) 526–535.
Kawakami K, Yoshikawa T, Moroto Y, Kanaoka E, Takahashi K, Nishihara Y., Microemulsion formulation for enhanced absorption of poorly soluble drugs I. Prescription design J Control Release, 81 (2002) 65-74.
Mei, Z.N., Chen, H.B., Weng, T., Yang, Y.J., Yang, X.L., Solid lipid nanoparticle and microemulsion for topical delivery of triptolide. Eur. J. Pharm. Biopharm. 56 (2003) 189–196.
Chang, X.L., Chen, H.B., Zhao, X.Z., Gao, Z.H., Xu, H.B., Yang, X.L., High- performance liquid chromatography determination of triptolide in vitro permeation studies Anal. Chim. Acta, 534 (2005) 215–221
Changez, M., Varshney, M., Chander, J., Dinda, A.K., Effect of the composition of lecithin/n-propanol/isopropyl myristate/water microemulsions on barrier properties of mice skin for transdermal permeation of tetracaine hydrochloride: in vitro. Colloids Surf. B: Biointerf. 50 (2006) 18–25.
Van Abbe N. Exaggerated exposure in topical irritancy and sensitization testing. J. Soc. Cosmet. Chem. 26 (1975) 173–187.