Evaluation of Dikamali as a Tablet Binder in Zidovudine Tablets
Keywords:
Dikamali, zidovudine, binder, natural gum, tabletAbstract
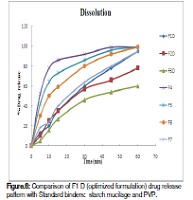
The aim of the present study is to evaluate the gum, Dikamali, as a tablet binder employing zidovudine as a model drug. Zidovudine tablets were prepared by wet granulation technique using Dikamali as a tablet binder. The Dikamali was used in wet form and dry form. Granules were evaluated for pre-compression parameters: tapped density, bulk density, compressibility index, hausner ratio, and angle of repose. All the parameters were found to be within the acceptable limits. The tablets were evaluated for hardness, friability, weight variation, disintegration, content uniformity, and dissolution. For the formulations F1-F3D; F1-F3W; F4-F7 (see Table 1) the parameters of friability, disintegration time, and hardness were measured and their values range from 0.57-0.73% (w/w), 0.83-0.97% (w/w), 0.69-0.99% (w/w); 12-13 min, 10-12 min, 10-12 min; and 5-6.9 kg/cm2, 4.5-5.1 kg/cm2, 4.1- 5.2 kg/cm2; respectively. The binding efficacy of Dikamali was compared with the standard binders, starch mucilage and polyvinyl pyrrolidone, using dissolution studies. The binders, Dikamali and starch, were compared at similar concentrations [2.5% (w/v), 5% (w/v), and 7.5% (w/v)], and the finalized formulation (F1D) was compared with a 10% (w/v) concentration of starch mucilage and a 10% (w/v) concentration of polyvinyl pyrrolidone (PVP). Dikamali [2.5% (w/v)] in dry form (i.e. F1D) showed the same percent drug release as that of the 10% (w/v) of starch mucilage and of polyvinyl pyrrolidone. In conclusion, Dikamali could well be used as a binding agent in the formulation of tablet dosage forms, and Dikamali is more effective in dry form than the wet form.
References
Oluremi BB, Bamiro OA, Idowu AO, Oduneye OA. Effect of compression pressure, preservative, and storage with potassium chloride on the microbiological quality of tablets formulated with Terminalia randii Gum (Combretaceae). Pak J Pharm Sci. 2012;25(4):773-776.
Prajapati ST, Patel MV, Patel CN. Preparation and evaluation of sublingual tablets of zolmitriptan. Int J Pharm Investig. 2014;4(1):27-31.
Suryawanshi SR, Thakare NP, More DP, Thombre NA. Bioavailability enhancement of ondansetron after nasal administration of Caesalpinia pulcherrima-based microspheres. Drug Deliv. 2013. [Epub ahead of print]
Jacques Scholtz, Jaco Van der Colff, Jan Steenekamp, Nicole Stieger and Josias Hamman. More Good News About Polymeric Plant- and Algae-Derived Biomaterials in Drug Delivery Systems. Curr Drug Targets. 2014;15(5):486-501.
Rahul V. Manek, Philip F. Builders, William M. Kolling, Martins Emeje, and Olobayo O. Kunle. Physicochemical and Binder properties of Starch obtained from Cyperus esculentus. AAPS PharmSciTech. Jun 2012; 13(2): 379–388.
Gavlighi HA, Michalak M, Meyer AS, Mikkelsen JD. Enzymatic depolymerization of gum tragacanth: bifidogenic potential of low molecular weight oligosaccharides. J Agric Food Chem. 2013;61(6):1272-1278.
Girish K Jania, Dhiren P Shahb, Vipul D Prajapatia, Vineet C Jainb. Versatile excipients for pharmaceutical formulations. Asian Journal of Pharmaceutical Sciences. 2009; 4 (5): 309-323.
Dr.K.S.Krishnan marg. Council of Scientific and Industrial Research. Wealth of India. New Delhi, India: CSIR Publishing house; 1956. 4. P.109-110.
Lakavath S, Avula B, Wang YH, Rumalla CS, Gandhe S, Venkat rao AR. Differentiating the gum resins of two closely related Indian Gardenia species, G. gummifera and G. lucida, and establishing the source of Dikamali gum resin using high-performance thin-layer chromatography and ultra-performance liquid chromatography-UV/MS. JAQAC Int, Pubmed. 2012; 95(1): 67-73.
C.P Khare (Ed.). Encyclopedia of Indian Medicinal Plants. New York, USA: Springer
science + Business media; 2007.P. 230–231.
Chopra RN. Indigenous Drugs of India. Kolkata, India: Bimal Kumar Dhur of academic
publishers; 2006.P.137.
Indian Council of Medical Research. Phytochemical Reference Standards of selected
indian medicinal plants. New Delhi, India: Medicinal Plants unit publisher; 2010.P.143-151.
Anandakumar A, muralidharan R, balasubramaniam M. Standardisation of Dikamali. Ancient Science of Life; 1984, 4(2): 106-109.
Kunert O, Sreekanth G, Babu GS, Rao BV, Radhakishan M, Kumar BR. Cycloartane triterpenes from Dikamali, the gum resin of Gardenia gummifera and Gardenia lucida. Chem biodivers (pubmed).2009; 6(8):1185-1192.
Santos JV, Pina ME, Marques MP, de carvalho LA. New sustained release of zidovudine matrix tablets - cytotoxicity toward Caco-2 cells. Drug Dev Ind Pharm. 2013 Aug; 39(8):1154-66.
Santos JV, Batista de carvalho LA, Pina ME. The influence of the compression force on zidovudine release from matrix tablets. AAPS Pharm Sci Tech. 2010 Sep; 11(3):1442-1448.
Martins EMEJE,* Olajide OLALEYE, Christiana ISIM. Oral Sustained Release Tablets of zidovudine Using Binary Blends of Natural and Synthetic Polymers. Biol. Pharm. Bull. 2010;33(9): 1561-1567.
H.P Rang, M.M.Dale, J.M.Ritter. Pharmacology. Spain: Elsevier publications; 2007.p.684-685.
Leon Lachman, Herbert A. Lieberman. The Theory and practice of Industrial Pharmacy.Bombay, India: Verghese publishing house; 1987.p.317-324.
Bettini R, Colombo P, Massimo G, Catellani PL, Vitali T. Swelling and drug release in hydrogel matrices: Polymer viscosity and matrix porosity effects. Eur J Pharm Sci. 1994; 2: 213-219.
Robert M. Silverstein, Francis X. Webster. Infrared Spectrometry. In: Robert M. Silverstein. editors. Spectrometric Identification of Organic Compounds. 6th Ed. John: Wiley and Sons. Inc. new York; 1997.p. 71 – 143.
Cooper J, Gunn C. Powder flow and compaction. In: Carter SJ,editors. Tutorial pharmacy. New Delhi: CBS publisher and Distributors; 1986.p. 211‐233.
Aulton ME, Wells TI. Pharmaceutics: The science of dosage form design. London, England: Churchill Livingstone; 1998.p.647‐649.
Government of India, Ministry of Health and Family Welfare. The Pharmacopoeia of India. Delhi, India: Controller of Publication; 5th Ed., Vol.II;2007.p.41-42.
Remington. The Science and Practice of pharmacy.19th Ed., Vol.I;1995.p.1669-1670.
Williams RL. (ed.) The United States Pharmacopoeia.29th ed. United States Pharmacopieal convention Inc., Rockville; 2006.p.958-959.