Formulation and evaluation of controlled porosity osmotic pump tablets of Glimepiride
Keywords:
Controlled porosity osmotic pump, glimepiride, glimepiridepore former, factorial designAbstract
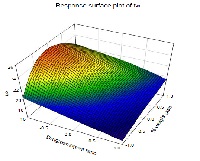
A Controlled porosity of the membrane is accomplished by the use of pore former. The usual dose of glimepiride was 4 mg to be taken twice daily. The plasma half life of glimepiride was 5 h. Hence, glimepiride was chosen as a model drug with an aim to develop a controlled release system for 24 h. Sodium chloride was use as osmogent. Cellulose acetate was used as the semi permeable membrane. The porous osmotic pump contains pore forming watersoluble additive (Poly ethylene glycol 400) in the coating membrane which after coming in contact with water, dissolve, resulting in an in situ formation of microporous structure. The effect of different formulation variables, namely, ratio of drug to osmogent, membrane weight gain and concentration of pore former on the in vitro release was studied using 23 full factorial design. The effect of pH and agitation intensity on drug release was also studied. It was found that drug release rate increased with the amount of osmogent because of increased water uptake. Drug release was inversely proportional to membrane weight gain. Surface plot is also presented to graphically represent the effect of independent variables on t90. Optimized formulation was found to release above 90% of glimepiride at a zero order rate for 24 h.
References
Rao BP, Geetha M, Purushothama N, Utpal S. Optimization and Development of Swellable Controlled Porosity Osmotic Pump Tablet for Theophylline. Tropical Journal of Pharmaceutical Research. 2009; 8: 247-255
Stuti G, Ravindra PS, Rohitashva S. Osmotic pumps: A review. International journal of comprehensive pharmacy. 2011; 2: 1-8.
Piyush G, Pankaj R, Dabeer A. A review on osmotically regulated devices. International journal of pharmacy & life sciences. 2010; 1: 302-312.
Drugs.com, Dec 2011, http://www.drugs.com/monograph/glimepiride.html
Chandra SY, Venu V, Jaganathan K. Formulation and in vitro evaluation of sustained release matrix tablets of glimepiride by using natural gums as release modifiers. Journal of Global Trends in Pharmaceutical Sciences. 2011; 2: 394-403.
Adel MA, Bassam IA. Preparation and Evaluation of Rapidly Disintegrating Glimepiride Tablets. International Journal of Pharmaceutical Sciences and Nanotechnology. 2011; 3: 1220-1229.
Parth P, Amish P, Tejas G, Vishal C. Formulation Development and Optimization of Controlled Porosity Osmotic Pump of Propranolol Hydrochloride. Journal of Pharmaceutics and cosmetology. 2011; 1: 7-14.
Patil S, Pund S, Joshi A, Shishoo CJ, Shahiwala A. Chronomodulated press coated pulsatile therapeutic system for aceclofenac: optimization of factors influencing drug release and lag time. Chrono Physiology and Therapy. 2011; 1: 1–10.
Patel GN, Patel RB and Patel HR. Formulation and in vitro evaluation of microbially triggered colon specific drug delivery using sesbania gum. 2011; 2: 6
Gennaro AR and Remington. The Science and Practice of Pharmacy, 19th edition, Mack Publishing Co., Eastern Pennsylvania, USA, 1995;1660:1676.
Lachman L. The Theory and Practice of Industrial Pharmacy, 3rd edition, 1987; 336, 413.
Gaylen MZ. Osmotic flow through controlled porosity films: An approach to delivery of water soluble compounds. Journal of Controlled Release. 1985, 2, 217-229.
Kanagale P, Lohray BB, Misra A. Formulation and Optimization of Porous Osmotic Pump–based Controlled Release System of Oxybutynin. AAPS PharmSciTech. 2007: 8.
Deepak G, Kilambi P. The Fabrication and Evaluation of the Formulation Variables of a Controlled Porosity Osmotic Drug Delivery System with Diltiazem Hydrochloride. Pharmaceutical technology. 2003: 58-68.
Rabiu Y, Kok KP, Yvonne TF. Design of a 24 h controlled porosity osmotic pump system containing PVP: formulation variables. Drug Development and Industrial Pharmacy. 2009; 35: 1430–1438.
Costa P, Sousa JM. Modeling and comparison of dissolution profiles. European Journal of Pharmaceutical Sciences. 2001; 13:123-133.
Higuchi T. Rate of Release of Medicament from Ointment bases containing drugs in Suspension. Journal of Pharmaceutical Science. 1961; 50: 874.
Korsemeyer RW, Ginnu R, Doelker E, Buri P, Peppas NA. Mechanism of Solute Release from Porous Hydrophilic Polymers. Journal of Pharmaceutics. 1983; 15: 25.