Formulation and Evaluation of Ketotifen Fumarate Fast Disintegrating Sublingual Tablets
Keywords:
Sublingual tablets, Ketotifen Fumarate, 2-HP-βCD, Fast disintegration, BioavailabilityAbstract
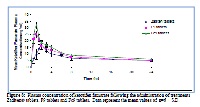
The aim of this study was to formulate KF fast disintegrating sublingual tablets (FDSLTs). KF undergoes first pass metabolism in liver, which has oral bioavailability of only 50% of the administrated dose. Sublingual dosage form bypasses the metabolism of KF in liver and offers a fast relieve of asthma. To achieve this goal, superdisintegrants and diluents were evaluated for their effect on the disintegration behavior of KF tablets. Full factorial design (24) was applied for a screening study in which four factors were used at two levels (low and high). The four factors, were the type of disintegrants either Ac-di-sol or Explotab, the concentration of each disintegrant (either 3% or 5% w/w), the binder either Avicel PH101 or PEG6000 and finally the diluent was either spray dried lactose or granular mannitol. The weight variation, content uniformity, friability, hardness, disintegration time, and in-vitro dissolution of the prepared formulae were evaluated. Hydroxy propyle betacyclodextrin (2-HP-β-CD) was used for increasing the absorption of KF tablets formulae. Solid binary system of KF with betacyclodextrin (2HP--β-CD) was prepared in molar ratios of 1:1 of drug to cyclodextrin. The formula F9 containing Ac-di-sol(5%w/w) , Avicel PH101(10%w/w) and granular mannitol as diluent was selected as best formula that has the least disintegration time of 20 seconds and the highest cumulative dissolution percent of 80.28% after the first minute. The relative bioavailability of F9 both with and without the cyclodextrin was compared. Zaditen® tablets were taken as reference standard. The formula F9 showed mean peak plasma concentration Cmax of 25.578±2.65ng/mL, the mean time of peak plasma concentration tmax of 1.167±0.258 h and the AUC (0-∞) of 319.894± 69.13ng.h/mL. However, FC9 that containing the drug in the form of a complex with 2HP-β CD showed Cmax of 30.901±4.963ng/mL, tmax of 1.083±0.186h and AUC (0-∞) of 382.166±55.278ng.h/mL. The results indicate that KF/2-HP-βCD fast disintegrating sublingual tablets may serve as a successful strategy for enhancing the bioavailability of KF drug.
References
Shojaei, A.H., Buccal mucosa as a route for systemic drug delivery: a review. J Pharm Pharm Sci., 1998. 1(1): p. 15-30.
El-Hawary, M., Khayal, M. and Issak, Z., in: “Handbook of Pharmacology”, . 1985. 377-384.
“Martindale; The Complete Drug Reference” ,Sean, C. Sweetman, . 2002, 33rd Ed.,The Pharmaceutical Press, London,. p.759-767.
“USP DI: Advice for the Patient Drug Information in Lay Language” Vol. II. 2001, 21st. Ed., MICROMEDEX, Thomson Healthcare. p. 920-921.
Yang, S., Fu, Y., Jeong, H.S., and Park, K., Application of polyacrilic superporous hydrogel microparticles as a sper-disintegrant in fast disintegrating tablets. J.P.P, 2004. 56: p. 429-436.
Del Valle, E.M.M., Cyclodextrins and their uses. a review. Process Biochemistry, 2004. 39: p. 1033-1046.
Challa, R., Ahuja, A., Ali, J. , and Khar, R.K., Cyclodextrins in drug delivery: an updated review. AAPS PharmSciTech , Article 34, 2005. 6(2): p. E329-E357.
Nagarsenker, M.S., Meshram, R.N. and Ramprakash, G., Solid dispersion of hydroxypropyl beta-cyclodextrin and ketorolac: enhancement of in-vitro dissolution rates, improvement in anti-inflammatory activity and reduction in ulcerogenicity in rats. J. Pharm. Pharmacol., 2000. 52(8): p.
-956.
Szejtli, J.a.S., L., Elimination of bitter, disgusting tastes of drugs and foods by cyclodextrins. Eur. J. Pharm. Biopharm, 2005. 61(3): p. 115-125.
Arima, H., et al., Comparative studies of the enhancing effects of cyclodextrins on the solubility and oral bioavailability of tacrolimus in rats. J Pharm Sci, 2001. 90(6): p. 690-701.
The Biritich Pharmacopoia. 2001: Vol. I and Il .London :The statiory office,. p.705-706,1810-1812
Ansel , H.C., Allen ,L.V. ,and Popovich ,N.G. , Pharmaceutical Dosage Form and Drug Delivery Systems". 7th ed. 1999: Philadelphia: Lippincott Wilkins ,Wolters Kluwer Company. p. 60-63.
Kottke, M.K. and E.M. Rudnic, Tablet Dosage Forms, in Modern Pharmaceutics, G.S. Banker, and Rhodes, C.T., Editor. 2002, Marcel Dekker Inc.: New York. p. 437-511.
Alderborn, G., Tablets and compaction, in Pharmaceutics The Science of Dosage Form Design, M.E. Aulton, Editor. 2002, Churchill Livingstone: Edinburgh. p. 397-439.
Abd El-Bary, A.A., Salem, H.F., Kharshoum, R.M., 2-Hydroxypropyl-ß-Cyclodextrin Complex with Ketotifen Fumerate for Eye Drops Preparations. International Journal of Drug Delivery, 2011. 3: p. 228-240.
Chiang, C.H., Chen, J.L., Liu, Y.T. , Therabutic effectband pharmacokineticsof ketotifen trans dermal delivary system. Drug Dev. Indust. Pharm., 1998. 24: p. 213-217.
Tapash, K., Ghosh and Pfister, W.R., Drug delivery to the oral cavity Molecules to the market. Taylor & Francis Group, 2005.
Conine, J.W., and Pikal.M.J., Pharmacutical Dosage FormsTablets, ed. H.A. Liberman, Lachman, L.,Schwartz J.B. Vol. 1. 1989: New York :Marcel Dekker. p.329-340.
Jacobsen, J., Christrup, L., and Jensen, N., Medicated chewing gum: pros and cons Am J Drug Delivery, 2004. 2(2): p. 75-88.
Seager, H., Drug-delivery products and the Zydis fast-dissolving dosage form. J Pharm Pharmacol,, 1998. 50(4): p. 375-382.
Loftsson, T., Jarho, P., Masson, M., and Jarvinen, T, Cyclodextrins in drug delivery. Exp. Opinion Drug Del, 2005. 2: p. 335-351.
Senel, S., and Hincal, A.A., Drug permeation enhancement via. buccal route: Possibilities and limitations. J. Control. Rel., 2001. 72: p. 133-144.
Vandecruys, R., Peeters, J., Verreck, G. and Brewster, M.E. , Use of a screening method to determine excipients which optimize the extent and stability of supersaturated drug solutions and application of this system to solid formulation design. Int. J. Pharm., 2007. 342(1-2): p. 168-175.
Yoo, S.D., Yoon, B.M., Lee, H.S. and Lee, K.C., Increased bioavailability of clomipramine after sublingual administration in rats. J. Pharm. Sci. , 1999. 88(11): p. 1119-1121.
Udata, C., Patel, J., Pal, D., Hejchman, E., Cushman, M. and Mitra, A.K., Enhanced transport of a novel anti-HIV agent--cosalane and its congeners across human intestinal epithelial (Caco-2) cell monolayers. Int. J. Pharm. , 2003. 250(1): p. 157-168.
Martins, P.S., Ochoa, R., Pimenta, A.M., Ferreira, L.A., Melo, A.L., da Silva, J.B., Sinisterra, R.D., Demicheli, C. and Frézard, F., Mode of action of beta-cyclodextrin as an absorption enhancer of the water-soluble drug meglumine antimoniate. Int. J. Pharm., 2006. 325(1-2): p. 39-47.